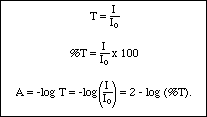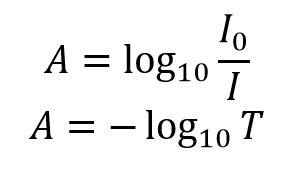Beer Lambert's Law, Absorbance & Transmittance - Spectrophotometry, Basic Introduction - Chemistry - YouTube

How can i calculate the band gap with absorbance or transmittance data using uv-vis spectroscopy? | ResearchGate
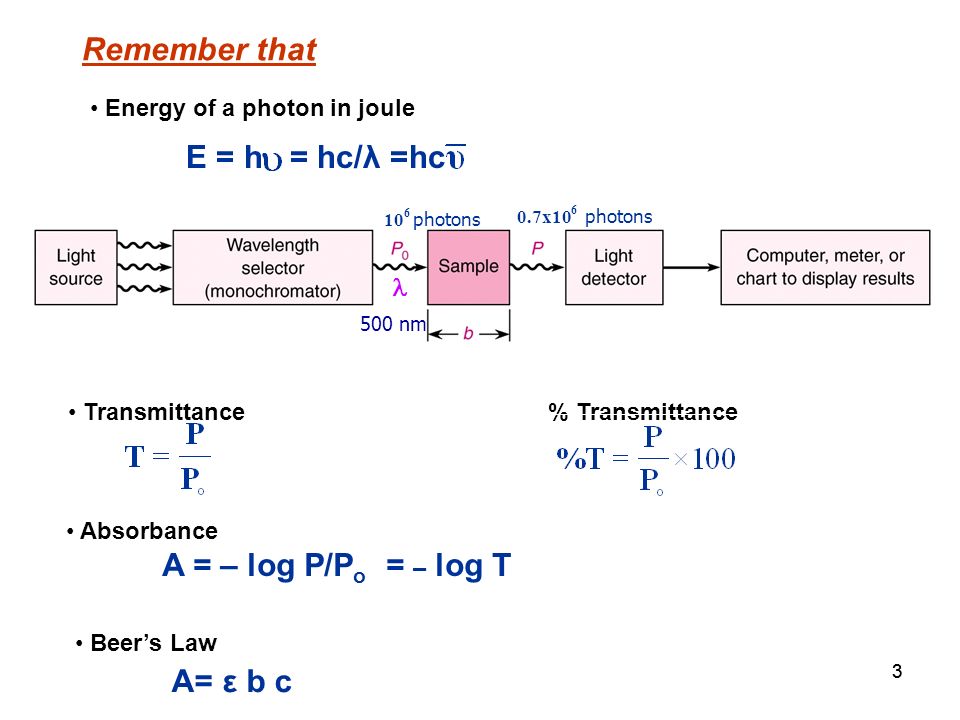
11 Instrumental Analysis Tutorial Use mathematical formulae to calculate absorbance, transmittance of a sample and wave parameters. Determine factors. - ppt download

SOLVED: The complex formed Cu(ll) and 1,10 Phenanthroline line has a molar absorptivity of 6.94x 103L cm-1 at 435nm; the wavelength of maximum absorption Calculate a) the absorbance of an 8.50x 10-5

Absorbance Transmittance| Numerical Practice problem on Lambert Beer Law| calculations and questions - YouTube

Absorbance Measurements – the Quick Way to Determine Sample Concentration - Eppendorf Handling Solutions